Understanding how plants control their rate of gas exchange and water loss is a fundamental part of plant biology. However, studying the tiny doughnut-shaped pores on leaves—known as stomata—using microscopy, can be time-consuming.
In two papers published last month in New Phytologist and Plant Methods, researchers from the University of Adelaide’s Waite Research Institute worked with the Australian Institute for Machine Learning to develop AI-powered software tools which can make analysing the dynamics of stomata less labour-intensive.
In both studies, researchers used a type of AI called deep computer vision to accurately determine the number of open and closed stomatal pores and their density on the leaf; as well as take precise measurements of a stomatal pore’s area, length, and aperture from microscope images.
Published in New Phytologist, software tool StomaAI was developed by machine learning PhD candidate James Bockman and Dr Na Sai during her plant physiology PhD.
Known to science since 1889, stomatal pores are vital to ecosystems as they act as gateways allowing carbon dioxide to enter a plant and oxygen and water to exit.
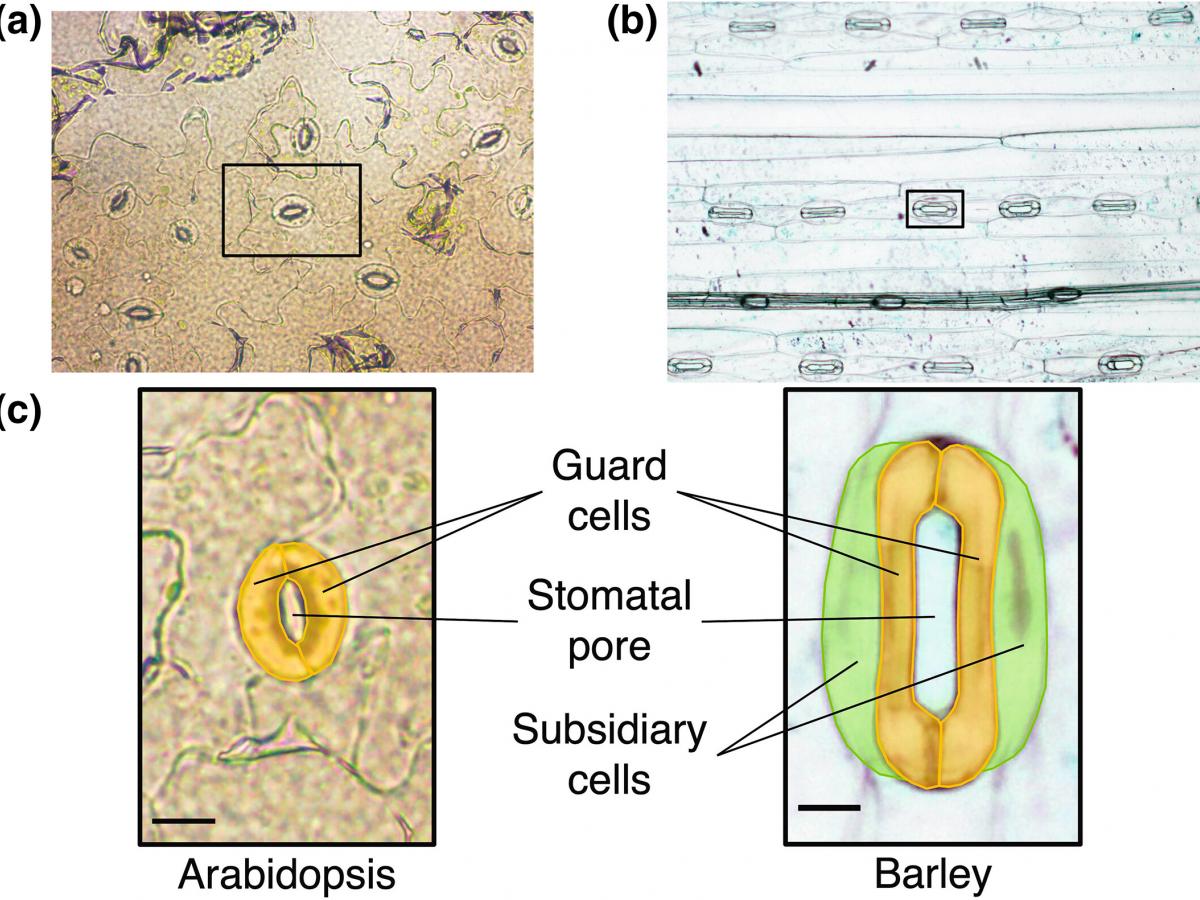
The AI software can accurately determine the number of open and closed stomatal pores and their density on the plant leaf in microscopy images.
Manually measuring the aperture of plant stomata allows researchers to get a clearer picture of how they function and respond to their environment. It is a common task for biologists studying plant signalling pathways and stress perception.
While some computer programs have attempted to semi-automate this process in the past, no such technology has allowed researchers to get results seconds after microscopy images are taken.
StomaAI has been initially trained to work on the crop plant barley and Arabidopsis, a favourite model organism of many plant biologists related to the crop plants canola, mustard, and broccoli. However, StomaAI can also be adapted by users to work on any other plant.
The strength of StomaAI is that it can study the signalling processes that control the dynamics of stomatal movement over minutes and hours, as it has been designed to work on isolated plant epidermis to which stimuli can easily be applied.
“The real beauty of this tool is that it takes so little time to get these measurements,” Dr Sai says.
“Usually, people take about 100 stomata apertures at a minimum, and that can take four hours to do, whereas we can get thousands of stomata and measure finer and finer effects in a fraction of the time using this tool.”
“It’s not just that you can speed up the analysis, it’s that it also instantly frees you up to change the kinds of experiments you’re doing.”
The benefit of the study published in Plant Methods is that it provides a fast, non-destructive way to measure the stomata on intact leaves using a handheld microscope.
It can be used in the field, in greenhouses, or labs to rapidly measure stomata density and aperture. The density and pore size of stomata in wheat, rice, and tomatoes were measured by senior author Dr Abdeljalil El Habti and his team.

The new software allows scientists to quickly measure stomata on intact plant leaves out in the field using a small handheld microscope.
Dr El Habti said the tool can quickly identify plants that are more tolerant of stresses, such as droughts.
“We can now characterise populations of thousands of plants in both the greenhouse and field, providing information that can help with the breeding of plants that use water efficiently.”
Both studies trained a deep neural network capable of multitasking so it could translate their biological measurement issue into certain vision tasks.
Through this research, Bockman—co-first author of StomaAI—says both groups wanted to show that it wasn’t just a machine learning problem they were looking to solve, but they wanted to test whether their creation could mimic the work of experts.
“Not only could we say it was really good at doing the computer vision task, but we were also saying it was as good as humans at doing the measuring task, which gives us assurance that it’s a drop-in replacement for a human doing it,” Bockman says.
The teams are now working to consolidate the tools, with help from the Australian Plant Phenomics Facility into an integrated software package.
‘StomaAI: an efficient and user friendly tool for measurement of stomatal pores and density using deep computer vision’ (2023) by Na Sai, James Paul Bockman, Hao Chen, Nathan Watson-Haigh, Bo Xu, Xueying Feng, Adriane Piechatzek, Chunhua Shen, and Matthew Gilliham, has been published in New Phytologist.
‘Rapid non-destructive method to phenotype stomatal traits’ (2023) by Phetdalaphone Pathoumthong, Zhen Zhang, Stuart J. Roy, and Abdeljalil El Habti has been published in Plant Methods.
This article was originally posted by the Australian Institute for Machine Learning (AIML) on the 17th April 2023.
